Charles Law Calculator
Charles Law Calculator is a online tool used for finding the ideal gas parameters in a isobaric process. All you have to do is enter the necessary inputs as per the input section of the tool and tap on the calculate button to get output i.e. final pressure or volume in no time.
Charle's Law Definition
Charle's Law also known as Law of Volumes denotes the relationship between volume and temperature when the mass, pressure of a gas are constant. Charles Law States that Volume and Temperature are directly proportional to each other.
In other words, we can frame the Charles Law Definition as Ratio of Volume and Temperature of a Gas in a closed system remains constant as long as the pressure is not changed. It denotes the behavior of an ideal gas in an isobaric process which indicates the pressure remains constant all through out this transition.
Charle's Law Formula
As per the definition of Charles Law we can write the equation as VΓéü / TΓéü = VΓéé / TΓéé where VΓéü and TΓéü are the intial volume and temperatures and VΓéé, TΓéé are the final values.
Let say you want to determine the final volume then the Charles Law Equation becomes VΓéé = VΓéü / TΓéü * TΓéé. On the other hand, if you want to set the final volume and estimate the temperature then rearranging the charles law formula we have TΓéé = TΓéü / VΓéü * VΓéé
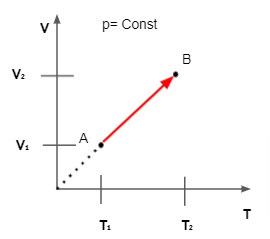
Charle's Law Derivation
As per Charles Law at Constant Pressure Volume of a gas is directly proportional to the Temperature. We can denote the statement as V∝T. We can remove the proportionality symbol and equate by placing a constant k i.e. V/T = Constant = k. The Constant k depends on the pressure of the gas as well as amount, units of volume, etc.
If we assume VΓéü and TΓéü as the initial volume and temperature at a constant pressure then the equation above can be written as VΓéü / TΓéü = k..... (1)
After a change in temperature its volume becomes VΓéé along with temperature TΓéé. We can write the equation as VΓéé / TΓéé = k...... (2)
Equating both the equations we get VΓéü / TΓéü = VΓéé / TΓéé or VΓéüTΓéé = VΓééTΓéü
Charles Law Examples
A sample of gas has an initial volume of 35L and an initial temperature of -45°C. What will be the temperature of the gas if the volume is 25L?
Solution:
Given that
Initial Volume (VΓéü) = 35L
Initial Temperature (TΓéü) = -45°C
Final Volume(VΓéé) = 25L
Final Temperature (TΓéé) =?
As per the Charles Law TΓéé = TΓéü / VΓéü * VΓéé
Substituting the known parameters in the equation we get the unknown parameter as TΓéé = -45°C/35*25 = -110.19°C
Physicscalc.Com has got concepts like friction, acceleration due to gravity, water pressure, gravity, and many more along with their relevant calculators all one under one roof.
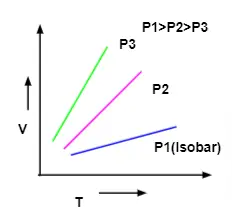
Charles Law Graph
Graph between Volume and Temperature at Constant Pressure is known as Isobar or Isoplestics and it always yields a straight line. A Plot of V Versus T at constant pressure is shown below for the temperature – 273.15°C and it is the lowest possible temperature.
Real Life Examples of Charles Law | Charle's Law Applications
There are various applications for Charles Law and we have compiled the most popular ones in the below sections.
- Balloon Flight: Have you ever seen a balloon fly in the sky and wondered how does it fly? It is because of Charles Law i.e. Whenever the air gets heated volume increases and density decreases. Bouyancy of Surrounding does the rest of the job for you and balloon begins to float.
- Liquid Nitrogen Experiments: If you place a balloon in a container filled with liquid nitrogen then it begins to shrink and freed no matter how it is at the initial stage. Later on, it gets back to its original stage after a temperature change and so is the volume.
- Thermometer: Thermometer is a device constructed based on the principle of Charle's Law. However, it has certain limitations like resistance to high temperatures, objects tensile strength, etc.
FAQs on Charles Law Calculator
1. What does Charles Law State?
Charles Law States that Volume of a Gas for fixed mass is directly proportional to temperature.
2. What are the Applications of Charles Law?
Charles Law is used in the areas of Balloon Flight, Liqid Nitrogen Experiments, Thermometer, several thermodynamic processes, etc.
3. What is the Formula for Charles Law?
Formula for Charles Law is given by the expression VΓéü / TΓéü = VΓéé / TΓéé
4. How to use the Charles Law Calculator?
Simply enter the initial volume, temperature and final volume or temperature and hit the calculate button to find out the other unknown parameter in the charles equation quite simply.
